SCIENCE & TECHNOLOGY
Vision for the Future
| Candidate | Indication | Targets | Preclinical | Clinical | IP |
|---|---|---|---|---|---|
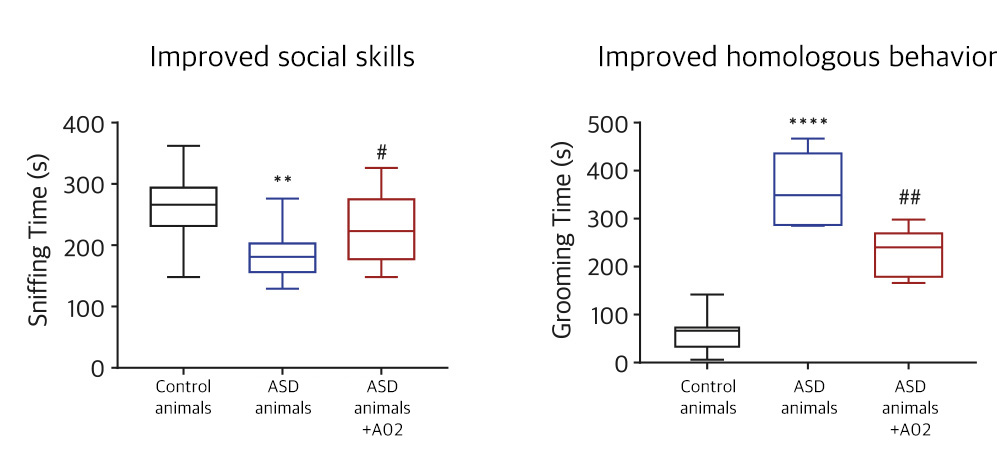
| NV01-A02 | Autism spectrum disorder | 5-HT Rc DA Rc |
Phase II (Korea)-LPO completed
|
KR, PCT, US, EU, CN, JP | |
| Fragile X syndrome |
Phase II (Global)-Preparation ongoing
|
KR, PCT, US, EU, CN, JP | |||
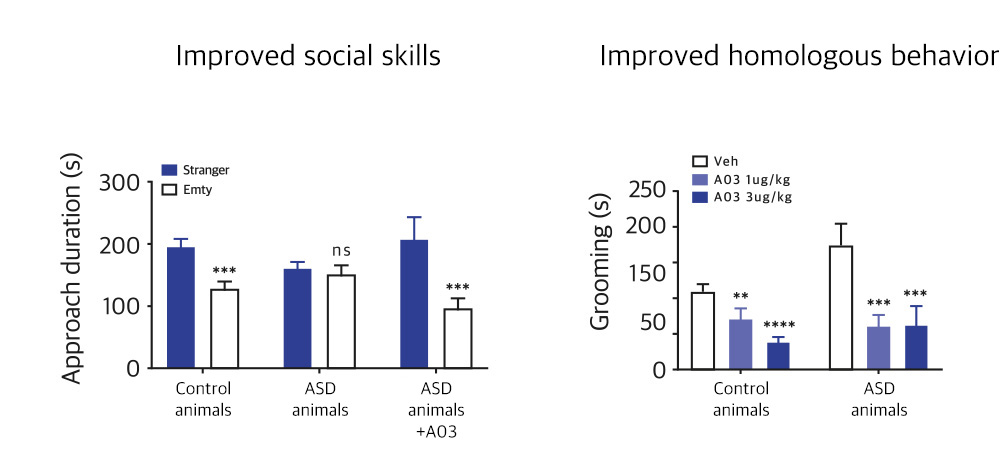
| NV01-A03 | Depression, Pain | Imidazoline Rc α-adrenergic Rc |
Phase II (Global)-Preparation ongoing
|
KR, PCT, US, EU, CN, JP | |
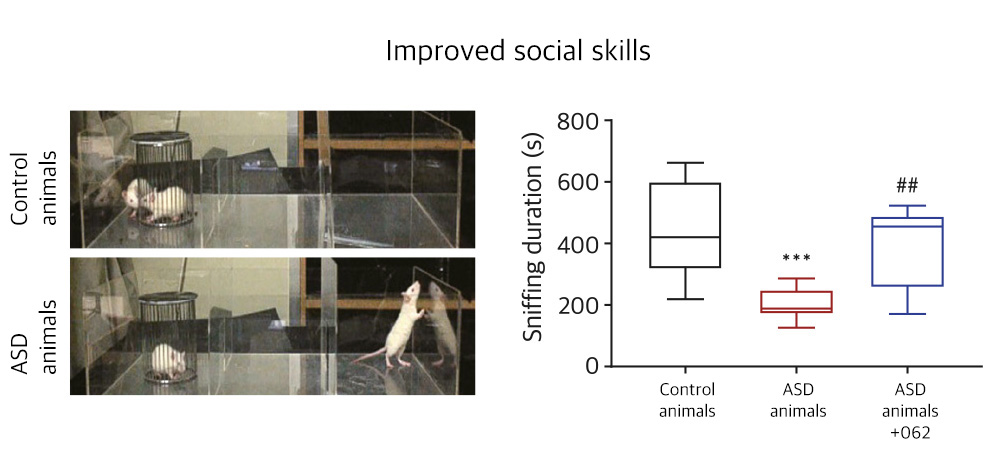
| NV01-062 | Depression, ASD | Imidazoline Rc NMDA Rc |
Proceeding
|
KR, PCT, US, EU, CN, JP | |
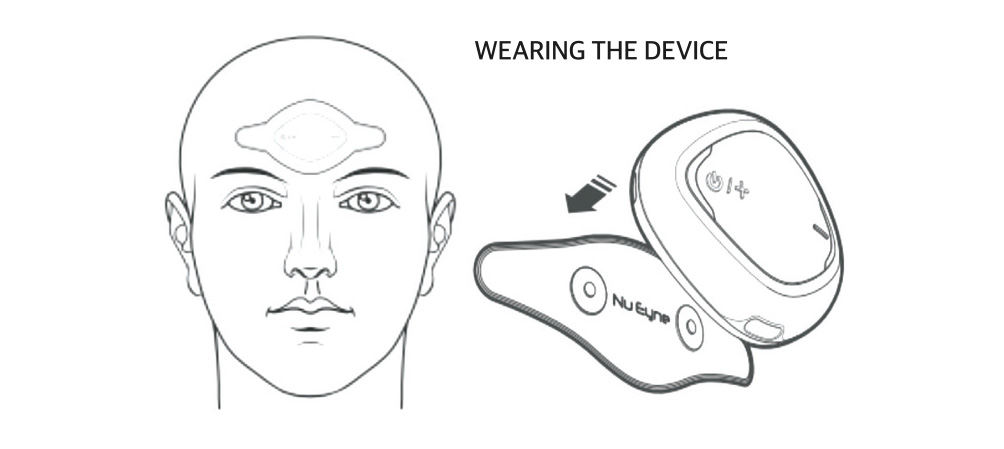
| NV01-E01 | Autism spectrum disorder | Electroceutical |
Confirmatory (Korea)-Preparation ongoing
|
- | |